Description
Usage:
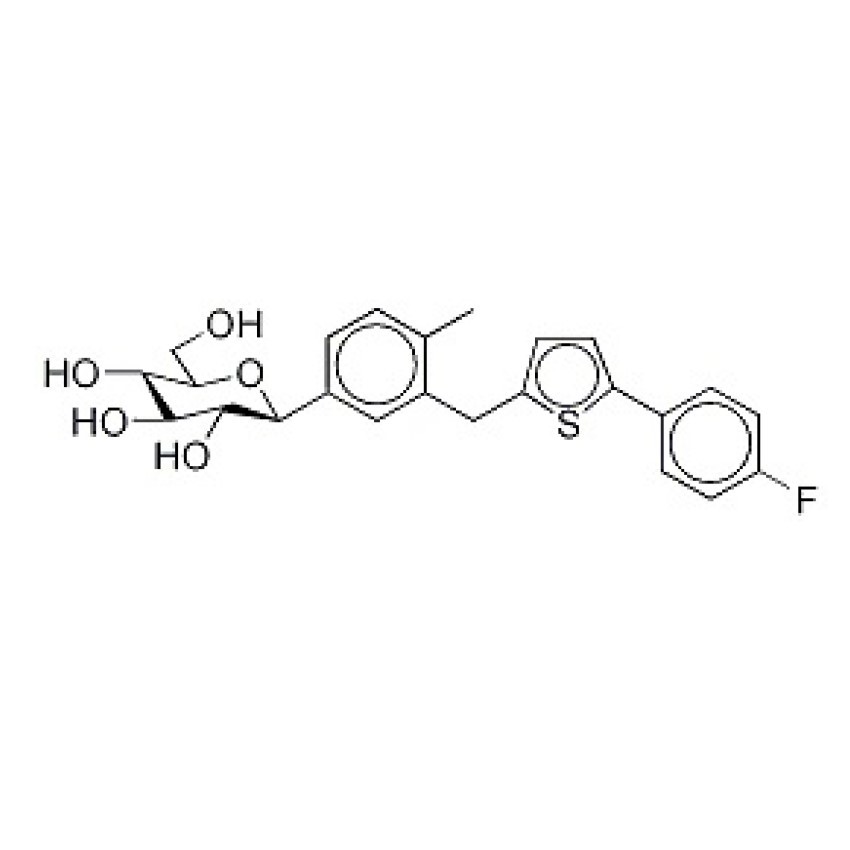
Canagliflozin(Invokana) is an oral diabetes medicine that helps control blood sugar levels. It works by helping the kidneys get rid of glucose from your bloodstream. Invokana is used together with diet and exercise to treat type 2 diabetes. To mention the advantage, this drug helps patients to reduce blood sugars without increasing the likelihood of gaining weight as long as a healthy, balanced diet is followed and regular exercise taken.
Canagliflozin(Invokana) is an oral diabetes medicine that helps control blood sugar levels. It works by helping the kidneys get rid of glucose from your bloodstream. Invokana is used together with diet and exercise to treat type 2 diabetes. To mention the advantage, this drug helps patients to reduce blood sugars without increasing the likelihood of gaining weight as long as a healthy, balanced diet is followed and regular exercise taken.
Canagliflozin is a sodium/glucose cotransporter 2 (SGLT2) inhibitor. Canagliflozin has been shown to dose dependently reduce calculated renal threshold for glucose excretion and increase urinary glucose excretion. Canagliflozin is a candidate for the treatment of type 2 diabetes and obesity.
| Product Name | Canagliflozin | ||
| Reference | USDMF standard | ||
| Test Items | Specifications | Results | |
| Appearance | White or almost white crystalline powder | White crystalline powder | |
| Identification | 1) Infrared absorption spectrum corresponds to the spectrum obtained with Canagliflozin RS. | Conforms | |
| 2) The retention time of the major peak in the chromatogram of the Sample solution corresponds to that in the chromatogram of the Standard solution, as obtained in the Assay. | Conforms | ||
| Water | Between 1.9% and 2.4% | 2.0% | |
| Residue on ignition | Not more than 0.1% | <0.1% | |
| Heavy metals | Not more than 0.002% | <0.002% | |
| Isomer(HPLC) | Not more than 0.15% | N.D | |
|
Related substancesl(HPLC) Furanose impurity Any other individual impurity Total impurities |
Not more than 0.15% Not more than 0.10% Not more than 1.0% |
N.D 0.05% 0.05% |
|
|
Related substances2(HPLC) Hydroperoxide impurity |
Not more than 200ppm | N.D | |
| Assay(HPLC) | 98.0% ~102.0% (Calculated on anhydrous and solvent-free basis) | 100.3% | |
|
Residual Solvents(GC) Methanol Ethyl acetate Tetrahydrofuran n-Heptane |
Not more than 3000 ppm Not more than 5000 ppm Not more than 720 ppm Not more than 5000 ppm |
N.D 460ppm N.D 44ppm |
|
| Conclusion | Complies with USDMF standard. | ||