Description
INTRODUCTION
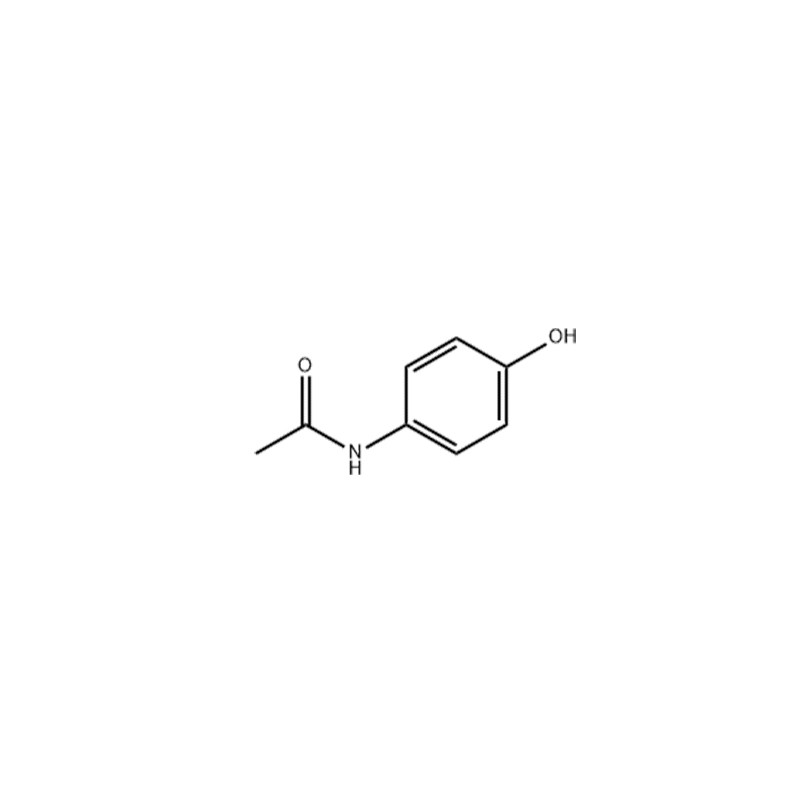
Acetaminophen (paracetamol) is a selective cyclooxygenase-2 (COX-2) inhibitor with an IC50 of 25.8 μM; is a widely used antipyretic and analgesic drug.
FUNCTION
1.Paracetamol is the most commonly used non-anti-inflammatory antipyretic analgesics, antipyretic effect similar analgesic effect is weak, no anti-inflammatory anti-rheumatic effects, acetanilide drugs is the best varieties. Particularly suitable for patients who can not use carboxylic acids. For cold, toothache embolism. Acetaminophen is also an intermediate for organic synthesis, hydrogen peroxide stabilizer, and photographic chemicals.
2.Paracetamol for antipyretic, analgesic, anti-rheumatic drugs
Acetaminophen (paracetamol) is a selective cyclooxygenase-2 (COX-2) inhibitor with an IC50 of 25.8 μM; is a widely used antipyretic and analgesic drug.
FUNCTION
1.Paracetamol is the most commonly used non-anti-inflammatory antipyretic analgesics, antipyretic effect similar analgesic effect is weak, no anti-inflammatory anti-rheumatic effects, acetanilide drugs is the best varieties. Particularly suitable for patients who can not use carboxylic acids. For cold, toothache embolism. Acetaminophen is also an intermediate for organic synthesis, hydrogen peroxide stabilizer, and photographic chemicals.
2.Paracetamol for antipyretic, analgesic, anti-rheumatic drugs
3.Paracetamol for organic synthesis intermediates, hydrogen peroxide stabilizer, photographic chemicals, non-anti-inflammatory antipyretic analgesics.
| Product name | Acetaminophen (Paracetamol) | ||||
| Quantity | 6000kgs | Packing | 25kgs /drum | ||
| Standard | BP2018/USP42 | ||||
| Contents | Specification | Results | |||
| Characters | White crystalline powder | Conforms | |||
| Identification | A:IR,UV,TLC Complies with the text for identification of Paracetamol | Conforms | |||
| B:Solubility : Sparingly soluble in water , freely soluble in alcohol, very slight soluble in methylene chloride . | Conforms | ||||
| Melting Point | 168-172℃ | 170.0-170.5℃ | |||
| Loss on drying | ≤0.5% | 0.07% | |||
| Residue on ignition | ≤0.1% | 0.04% | |||
| Heavy metals | ≤10ppm | <10ppm | |||
| Free p-aminophenol | ≤0.005% | Below LOD | |||
| Related substances | Impurity B | ≤0.05% | Below LOD | ||
| Impurity C | ≤0.05% | Below LOD | |||
| Impurity D | ≤0.05% | Below LOD | |||
| Impurity J | ≤0.001% | Below LOD | |||
| Individual unspecified impurity | ≤0.05% | 0.03% | |||
| Total of impurities | ≤0.1% | 0.04% | |||
| Assay(anhydrous basis) | 99.0%-101.0% | 99.70% | |||
| Conclusion: It complies with the requirements of the BP2018/USP42 | |||||