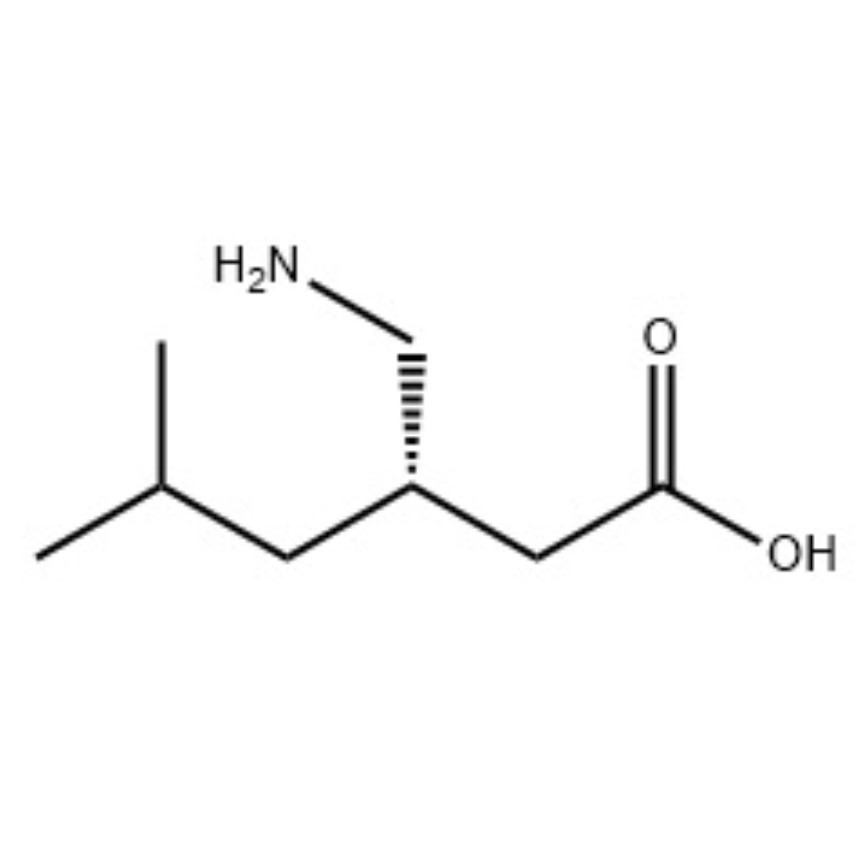
Description
USAGE:
· Management of neuropathic pain associated with diabetic peripheral neuropathy
· Management of postherpetic neuralgia
· Adjunctive therapy for the treatment of partial-onset seizures in patients 1 month of age and older
· Management of fibromyalgia
· Management of neuropathic pain associated with diabetic peripheral neuropathy
· Management of postherpetic neuralgia
· Adjunctive therapy for the treatment of partial-onset seizures in patients 1 month of age and older
· Management of fibromyalgia
· Management of neuropathic pain associated with spinal cord injury
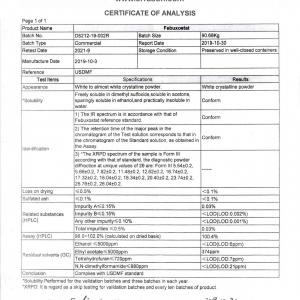
| Product Name | Pregabalin | ||
| Reference | USP | ||
| Test Items | Specifications | Results | |
| Appearance | White to almost white crystalline powder. | White crystalline powder | |
| Identification | A) IR spectrum is accordant with that of reference standard. | Conform | |
| B) The retention time of the major peak of the sample solution corresponds to that of the standard solution, as obtained in the Assay. | Conform | ||
| Loss on drying | ≤0.5% | 0.1% | |
| Residue on ignition | ≤0.1% | <0.1% | |
| Chloride | ≤0.1% | <0.1% | |
| Related substances (HPLC) | USP related compound C≤0.15% | <LOD(LOD:0.004%) | |
| Isobutylglutaric acid(PGB-3)≤0.15% | <LOD(LOD:0.02%) | ||
| lsobutylglutarmonoamide(PGB-5)≤0.15% | <LOD(LOD:0.02%) | ||
| Any unspecified impurity≤0.10% | <LOQ(LOQ:0.05%) | ||
| Total impurities≤0.5% | <LOQ | ||
| Isomer (HPLC) | USP related compound A≤0.15% | 0.04% | |
| Assay (HPLC) | 98.0-102.0% (dried basis) | 99.6% | |
| Residual solvents(GC) | Ethanol≤5000 ppm | <LOD(LOD:3.6ppm) | |
| lsopropanol≤5000 ppm | 320ppm | ||
| Chloroform ≤60 ppm | <LOD(LOD:3.5ppm) | ||
| Conclusion | Complies with USP | ||