Description
INTRODUCTION
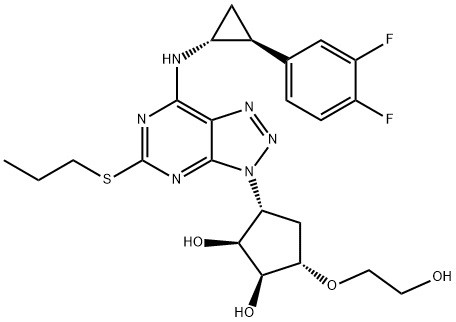
Ticagrelor is a platelet aggregation inhibitor produced by AstraZeneca. Unlike clopidogrel, ticagrelor is not a prodrug and does not require metabolic activation. Ticagrelor is an antagonist of the P2Y12 receptor.
Ticagrelor was approved for use in the European Union by the European Commission on December 3, 2010. The drug was approved by the US Food and Drug Administration on July 20, 2011.
Function
1. Ticagrelor is used to lower the chance of heart attack, stroke, and death in some people.
Ticagrelor is a platelet aggregation inhibitor produced by AstraZeneca. Unlike clopidogrel, ticagrelor is not a prodrug and does not require metabolic activation. Ticagrelor is an antagonist of the P2Y12 receptor.
Ticagrelor was approved for use in the European Union by the European Commission on December 3, 2010. The drug was approved by the US Food and Drug Administration on July 20, 2011.
Function
1. Ticagrelor is used to lower the chance of heart attack, stroke, and death in some people.
2. Ticagrelor is used to lower the chance of blockage of a stent after a stent is placed in the heart.
| Product name | Ticagrelor |
| Appearance | White powder |
| Density | 1.67g/cm3 |
| Loss on drying | ≤0.5% |
| Residue on ignition | ≤0.5% |
| Single impurity | ≤0.5% |
| Total impurities | ≤1.0% |
| Assay | ≥99% |
| Heavy metals | <20ppm |
| Ethanol | <5000ppm |
| N(CH3CH2) | <300ppm |