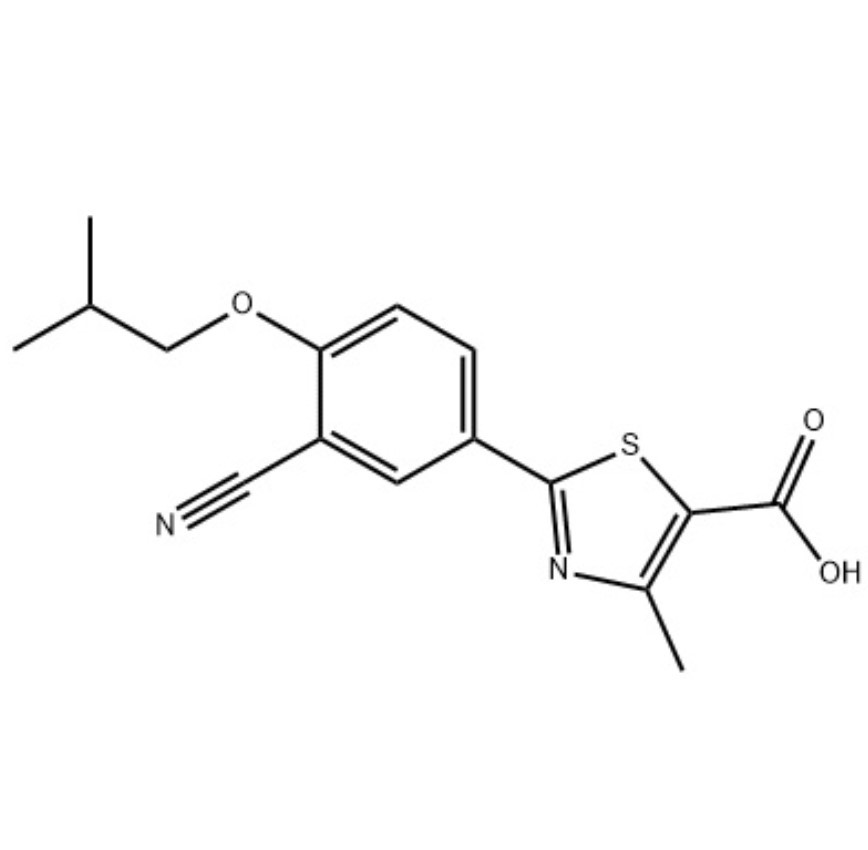
Description
Febuxostat is a xanthine oxidase inhibitor, clinically used to treat hyperuricemia (gout). It is a new and highly effective selective inhibitor of non-purine xanthine oxidase. Xanthine oxidase is the key enzyme to promote uric acid production. Febuxosutan can reduce the uric acid level in the blood of patients with hyperuricemia and gout. Clinical studies have shown that the drug is safe and effective. The drug is metabolized through the liver and does not rely on renal excretion. Doses of 40 mg or 80 mg once daily are not recommended for gout patients without hyperuricemia.
Function and Application
1.Febuxostat(TEI 6720;TMX 67 ) is selective xanthine oxidase inhibitor with Ki of 0.6 nM.IC50 value: 0.6 nM (Ki)
2.Febuxostat (INN; trade names Adenuric [EU], Febutaz [India] and Uloric [US]) is a urate lowering drug, an inhibitor of xanthine oxidase that is indicated for use in the treatment of hyperuricemia and chronic gout.
3.Febuxostat reduces the production of uric acid in your body. Uric acid buildup can lead to gout or kidney stones.
4.Febuxostat is used to treat gout.
5.Febuxosta ,is a anti-gout Pharmaceutical Raw Materials.
| Product Name | Febuxostat | ||
| Reference | USDMF | ||
| Test Items | Specifications | Results | |
| Appearance | White to almost white crystalline powder. | White crystalline powder | |
| *Solubility | Freely soluble in dimethyl sulfoxide.soluble in acetone, sparingly soluble in ethanol,and practically insoluble in water. | Conform | |
| Identification | 1) The IR spectrum is in accordance with that of Febuxostat reference standard. | Conform | |
| 2) The retention time of the major peak in the chromatogram of the Test solution corresponds to that in the chromatogram of the Standard solution, as obtained in the Assay. | Conform | ||
| 3) *The XRPD spectrum of the sample is Form III according with that of standard, the diagnostic powder diffraction at unique values of 2Θ are: Form III 5.54±0.2, 5.66±0.2, 7.82±0.2, 11.48±0.2, 12.62±0.2, 16.74±0.2, 17.32±0.2, 18.04±0.2, 18.34±0.2, 20.40±0.2, 23.74±0.2, 25.76±0.2, 26.04±0.2. | — | ||
| Loss on drying | ≤0.5% | <0.1% | |
| Sulfated ash | ≤0.1% | <0.1% | |
| Related substances (HPLC) | Impurity A≤0.15% | 0.03% | |
| Impurity B≤0.15% | <LOD(LOD:0.002%) | ||
| Any other impurity≤0.10% | <LOD(LOD:0.001%) | ||
| Total impurities ≤0.5% | 0.03% | ||
| Assay (HPLC) | 98.0~102.0% (calculated on dried basis) | 100.4% | |
| Residual solvents (GC) | Ethanol≤5000ppm | <LOD(LOD:6ppm) | |
| Ethyl acetate≤5000ppm | 374ppm | ||
| Tetrahydrofuran≤720ppm | <LOD(LOD:7ppm) | ||
| N,N-dimethylformamide≤880ppm | <LOD(LOD:21ppm) | ||
| Conclusion | Complies with USDMF standard | ||