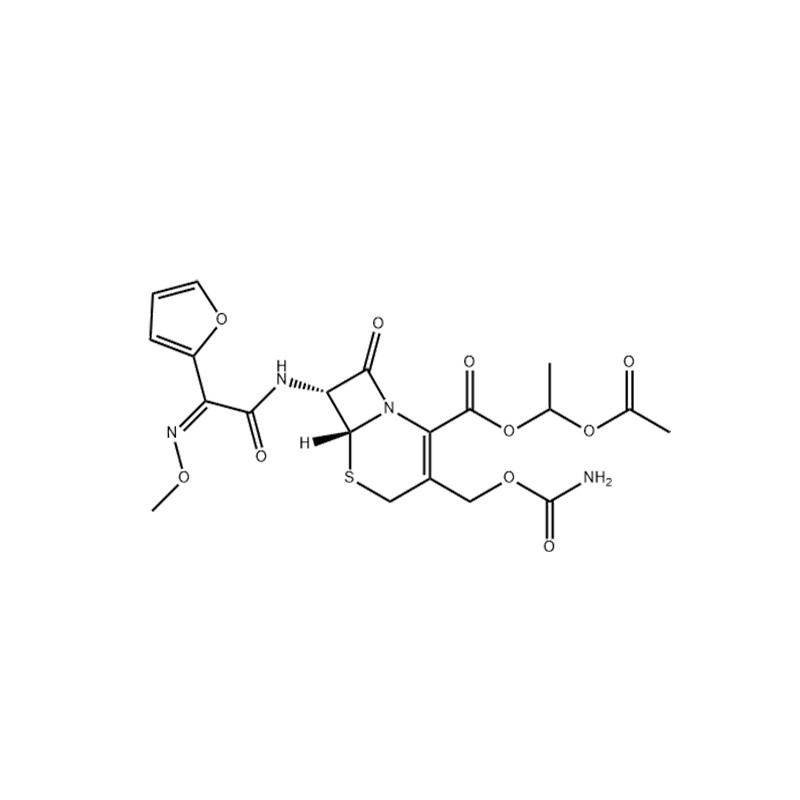
Description
What is cefuroxime axetil?
Cefuroxime is the second-generation broad-spectrum semi-synthetic cephalosporin, and it has almost all antibacterial effects on all strains that are sensitive to cefalotin and ceftazidime, and it is also effective against a wide range of gram-negative bacilli including enterobacteriaceae and sputum Proteus is effective.
Function of cefuroxime axetil
Cefuroxime is the second-generation broad-spectrum semi-synthetic cephalosporin, and it has almost all antibacterial effects on all strains that are sensitive to cefalotin and ceftazidime, and it is also effective against a wide range of gram-negative bacilli including enterobacteriaceae and sputum Proteus is effective.
Function of cefuroxime axetil
Cefuroxime clinical treatment of respiratory tract, facial features, bone, soft tissue infections and meningitis, simple urinary tract infection clinical and bacteriological effective rate of 70% to 100%, prevention of postoperative wound infection also has a satisfactory effect. Cefuroxime is effective in the treatment of acute gonorrhea and urinary tract infections caused by gonococcal infection. A hospital in 169 cases of gonorrhea patients, intramuscular injection of cefuroxime a single dose of 1.5g, 160 cases of intramuscular injection disappeared in 1 to 5 days, the cure rate was 97.7%, short-term and long-term bacteriological cure rate of cefuroxime 55% and 48% .
| Product name | Cefuroxime axetil | ||
| Packing | 20kgs/drum | Batch Size | 200kgs |
| Standard | USP42 | ||
| Contents | Specification | Results | |
| Characters | White or almost powder | Conforms | |
| Identification | IR | Conforms | |
| HPLC | Conforms | ||
| Crystalline | Amorphous | Conforms | |
| Diastereoisomer ratio | 0.48-0.55 | 0.54 | |
| Water | ≤1.5% | 0.26% | |
| Organic impurities | Methoxyiminofuranyl acetic acid a ≤0.30% | Not detected | |
| Cefuroxime b ≤0.50% | Not detected | ||
| Cefuroxime lactone c≤0.30% | Not detected | ||
| Cefuroxime axetil △-3isomersd,e≤1.20% | Not detected | ||
| Cefuroxime axetil E-isomersd, f.≤1.00% | 0.18% | ||
| Cefuroxime axetil dimerg.h≤0.50% | Not detected | ||
| Any other individual impurity≤0.30% | Not detected | ||
| Total impurites≤3.0% | 0.18% | ||
| Organic volatile impurities | Acetone ≤0.5% | 0.13% | |
| Ethyl acetate ≤0.5% | 0.03% | ||
| Isopropanol ≤0.5% | Not detected | ||
| Assay | 745-875ug/mg | 836ug/mg | |
| Conclusion: It complies with the requirements of the USP42 | |||